Mean Kinetic Temperature (MCT) is an internationally accepted parameter for establishing storage conditions for medicines.
by Eng. David Ernesto Preza Hernández*
TCM is then defined and its importance is discussed, as well as its differences with average temperature. An explanation is also made about the calculation of the same and the realization of temperature mappings to find critical points in a storage facility.
I. Introduction: One of the most important aspects to consider when storing medicines is temperature, as this directly impacts the stability, quality and shelf life of products. The same product can have different shelf life times depending on the environmental conditions under which it is stored. For example, a drug manufactured to be marketed in Canada will have a different shelf life than if it were marketed in Costa Rica.
The stability of the storage conditions is of great importance, since a change of just a few degrees Celsius can cause a product to shorten its shelf life by months or even years, as shown in graph # 1 in which we can see that an increase of 4 degrees decreases the shelf life of the product by 1 year, which, if not considered, becomes a health hazard and a considerable economic loss, a current issue where you can appreciate the importance of a well-designed cold chain, is for the storage of Covid 19 vaccines and the reagents necessary to perform the various Covid 19 Tests.
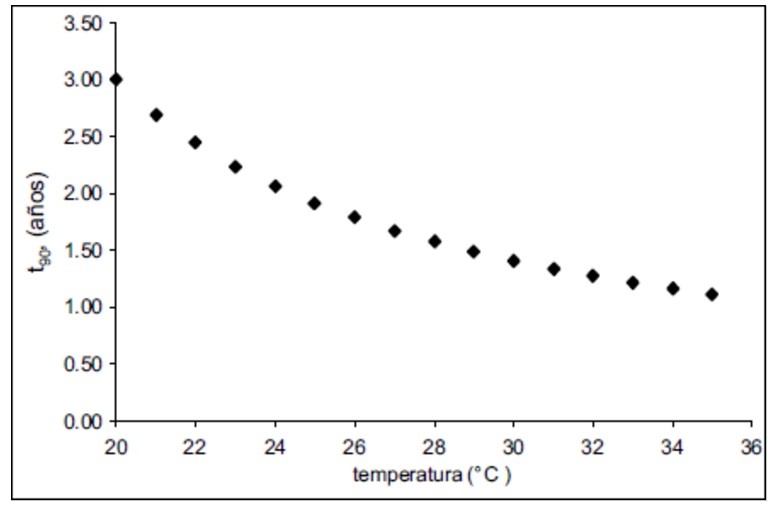
Graph #1 Variation of service life with storage temperature [2]
For this reason it is of great importance to establish and control the storage conditions for such products, by means of refrigeration systems or air conditioning systems.
Throughout my professional experience I have been able to show that in many drug storage warehouses the designers of these spaces are unaware of this parameter of great importance when establishing the central temperature of the system and the maximum and minimum tolerance limits.
In addition, in warehouses or facilities that are already built and that you want to adapt to store medical products, a mapping (study) must be carried out to determine the points of greatest criticality (coldest points and hottest points), in order to carry out actions that guarantee the quality of the products.
I. Mean Kinetic Temperature (Tcm)
Definition: According to the U. S. Pharmacopeia (USP 35 Chapter <1150>, "Pharmaceutical Stability"), it defines TCM as a calculated temperature value in which the total degradation produced, in a given period of time, is equivalent to the sum of the individual degradations that occur at different temperatures.
Another definition is that provided by the ICH (International Conference on Harmonization) Q1A (R2) "Stability Testing of New Drug Substances & Products, which define TCM as the temperature that, maintained for a certain time, produces the same thermal challenge to a product similar to that which it would have suffered if it were subjected to a temperature range between a maximum and a minimum in a certain period of time.
In other words, we can define the TCM as a temperature value which considers all the temperature changes that the product undergoes in a period of time. This concept should not be confused with a simple arithmetic average, since TCM considers the exponential increase in the degradation of materials as a function of temperature, so it establishes a non-linear relationship of the effect of temperature.
The TCM equation was developed by Jens T. Carstensen, among others. And summarizing it can be calculated using the following equation:

Figure 2.
Pharmaceuticals are designed to be stored at a certain TCM value, as can be intuited from the above definitions, TCM is largely related to the thermal stability of the storage area, hence air conditioning systems must be designed in such a way that they maintain a stability that is preserved within the appropriate tolerance limits to obtain the expected TCM value.
III. Temperature mapping
Definition: The World Health Organization in the technical guide "Technical Report Series, No. 961, 2011", says that a temperature mapping should be carried out in all those areas where products that have "storage temperature" vignettes are stored or handled, within these areas are cold rooms, refrigerators, temperature controlled areas and the like.
Temperature mappings are performed in order to locate the hottest and coldest spots in an area, as well as locate locations where temperatures are outside the established limits, all in order to perform corrective actions if necessary.
Any organization assigned the task of designing an air conditioning system must be aware that it is not enough to calculate the thermal load; thermal stability must be guaranteed within the limits that the client requires, in addition to ensuring that all
areas where the product is located are within these limits, and the only way to achieve this is by mapping the area.
To perform a temperature mapping, certain requirements must be followed and met:
• Generate a protocol (guide) where the requirements of the area to be mapped are established, for example the temperature values in which the area can vary (-25.0°C to -10.0°C, 2.0°C to 8.0°C, 15.0°C to 25.0°C, etc.).
• A sufficient number of Data loggers condition storage devices must be possessed to cover the entire area of interest, which must be programmed with a sampling time between 1 minute and a maximum of 15 minutes.
• These must have a calibration certificate with current traceability and with at least 3 calibration points within the range of interest of the mapping; an error of no more than ± 0.5°C must be guaranteed.
• The area where the mapping will be carried out must be described, denoting the dimensions, type of shelves, heights of the same, in addition to placing the sources of heat or cold that may affect the measurements.
• Determine the location of data loggers. This part is one of the most important of the process since a bad selection of locations can yield unrepresentative data. The personnel in charge of carrying out the mapping must have a great experience to determine the positions in which possible non-compliance with the requirements can be found.
For example, in storage structures such as the one shown in Figure #1, data loggers can be located at 3 different heights trying to cover as much area as possible and ensure measurement in all possible product locations.
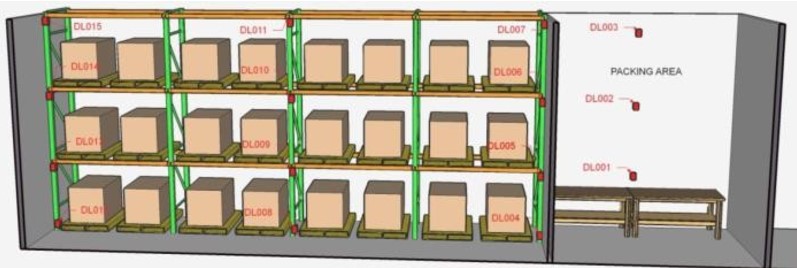
Figure #1 Typical Location of Data Logger Sensors
The WHO technical guide No. 961, 2011 of an example in which in a structure whose height is greater than 3.6 m the data loggers must be placed at 0.3 m, 1.8 m, 3.6 m and 5.4 m, in order to have adequate readings.
• The temperature mapping must be carried out in a period of time that depends on the type of installation for areas such as warehouses or warehouses, the usual thing is that the sampling period is 7 days, it is intended to observe the behavior of the area in work and rest periods, for temperature controlled facilities such as cold rooms the usual period is 24 or 72 hours.
• The data must be downloaded and the graphs obtained analyzed, an example of them can be seen in Figure # 2, of these graphs it is mainly interesting to note those points in which the equipment left conditions, to analyze the reason for that failure and perform the necessary corrective actions.
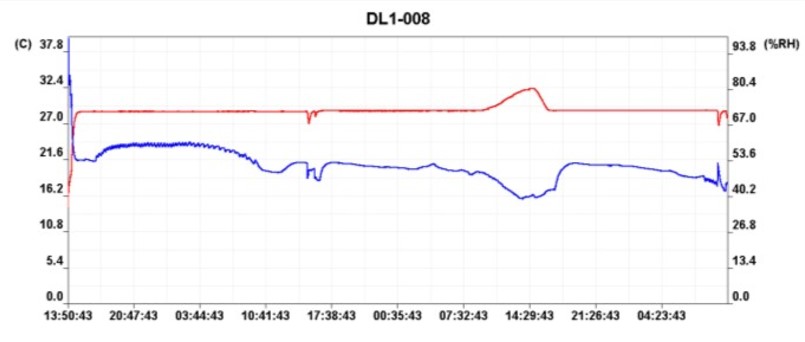
Figure # 2 Graph temperature and humidity Vrs Time.
References
[1] VAISALA Application note Mean Kinetic Temperature in GxP Environments
[2] GALLARDO C., Cecilia; ROJAS C., Jhon J.; FLÓREZ A., Oscar A. - The Mean Kinetic Temperature in studies of long-term stability and storage of medicines - Vitae, vol. 11, no. 1, 2004, pp. 67-72 University of Antioquia.
[3] World Health Organization Technical Report Series, No. 961, 2011
[4] U.S. Pharmacopeia (USP 35 Charter) "Pharmaceutical Stability"
 * Eng. David Ernesto Preza Hernández - [email protected] Tel: +50377781917
* Eng. David Ernesto Preza Hernández - [email protected] Tel: +50377781917















